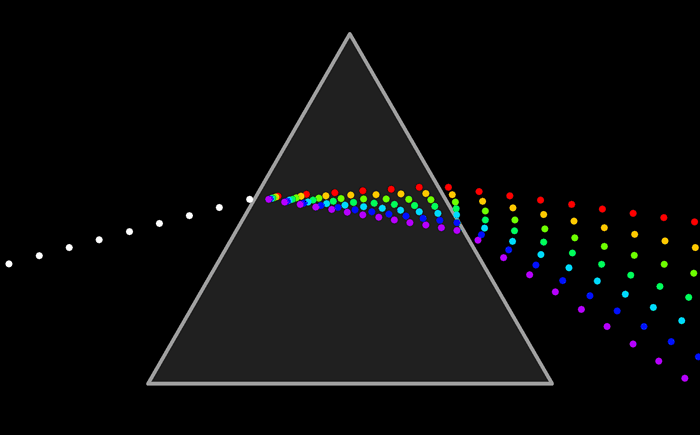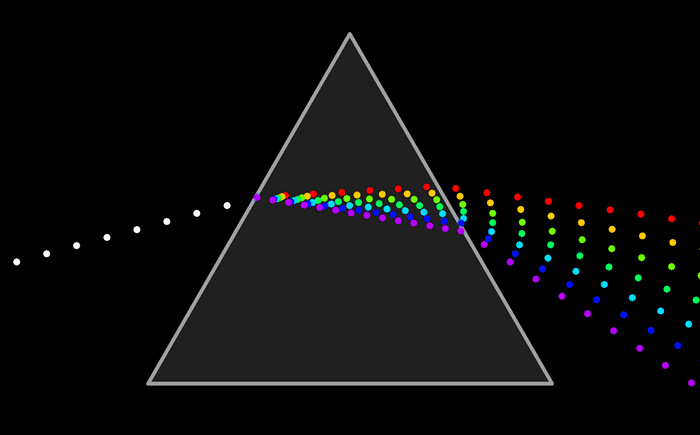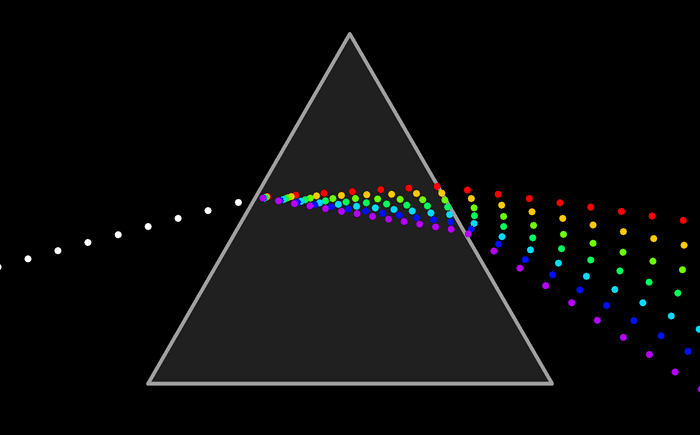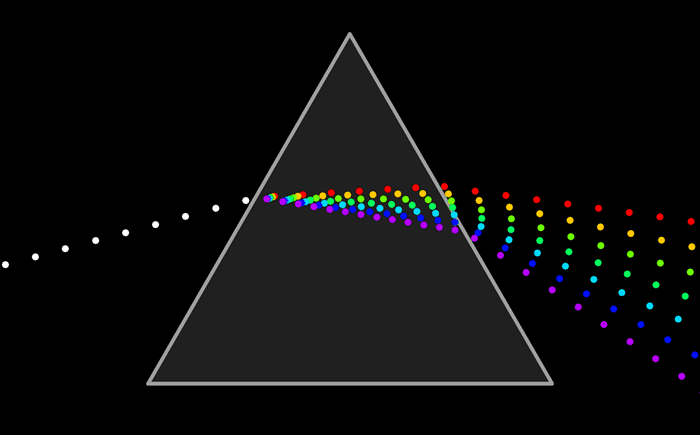
 Spectroscopy was originally the study of the interaction between radiation and matter as a function of wavelength (λ). In fact, historically , spectroscopy referred to the use of visible light dispersed according to its wavelength, e.g. By a prism. Later the concept was expanded greatly to comprise any measurement of a quantity as a function of either wavelength or frequency. Thus it also can refer to a response to an alternating field or the varying frequency (ν). Spectroscopy originated through the study of visible light dispersed according to its wavelength. Later the concept was expanded greatly to comprise any interaction with radiative energy as a function of its wavelength or frequency. A further extension of the scope of the definition added energy (E) as a variable, once the very close relationship E = hν for photons was realized (h is the Planck constant). A plot of the response as a function of wavelength—or more commonly frequency—is referred to as a spectrum.
Spectroscopy was originally the study of the interaction between radiation and matter as a function of wavelength (λ). In fact, historically , spectroscopy referred to the use of visible light dispersed according to its wavelength, e.g. By a prism. Later the concept was expanded greatly to comprise any measurement of a quantity as a function of either wavelength or frequency. Thus it also can refer to a response to an alternating field or the varying frequency (ν). Spectroscopy originated through the study of visible light dispersed according to its wavelength. Later the concept was expanded greatly to comprise any interaction with radiative energy as a function of its wavelength or frequency. A further extension of the scope of the definition added energy (E) as a variable, once the very close relationship E = hν for photons was realized (h is the Planck constant). A plot of the response as a function of wavelength—or more commonly frequency—is referred to as a spectrum.Spectrometry is the spectroscopic technique used to assess the concentration or amount of a given species. In those cases, the instrument that performs such measurements is a spectrometer or spectrograph.
Spectroscopy/spectrometry is often used in physical and analytical chemistry for the identification of substances through the spectrum emitted from or absorbed by them. Spectroscopy/spectrometry is also heavily used in astronomy and remote sensing. Most large telescopes have spectrometers, which are used either to measure the chemical composition and physical properties of astronomical objects or to measure their velocities from the Doppler shift of their spectral lines.
replica Sunglasses UK, combining elegant style and cutting-edge technology, a variety of styles of replica ysl Sunglasses, the pointer walks between your exclusive taste style.
ReplyDelete