|
Nanoparticle type
|
Applications
|
|
Alumino silicate nano particles
|
It can be used to reduce bleeding in trauma
patients with external wounds by activating the blood clotting mechanism,
causing blood in a wound to clot quickly. Z-Medica is producing a medical
gauze that uses aluminosilicate nano particles for use on external wounds. For
trauma patients with internal bleeding another way to reduce the blood loss
is needed.
Also the researchers at Chase Western Reserve University are developing polymer
nanoparticles that act as synthetic platelets. Lab tests have shown that injection of these synthetic platelets
significantly reduces blood loss.
|
|
Polyethylene glycol-hydrophilic carbon clusters
(PEG-HCC)
|
They have been shown to absorb
free radicals at a much higher
rate than the proteins out body uses for this function. This ability to
absorb free radicals may reduce the harm that is caused by the release of
free radicals after a brain injury.
|
|
Iron oxide nano particles
|
It can be used to improve MRI images of cancer tumours.
The nanoparticle is coated with a peptide that binds to a cancer tumour, once
the nanoparticles are attached to the tumour the magnetic property of the iron oxide enhances the images from the Magnetic
Resonance Imagining scan.
|
|
Gold nano particles
|
A method being developed to fight skin cancer
uses gold nanoparticles to which RNA molecules are attached. The
nanoparticles are in an ointment that is applied to the skin. The nanoparticles
penetrate the skin and the RNA attaches to a cancer related gene, stopping the gene from generating proteins that are used in the
growth of skin cancer tumours.
|
|
Gold nano particles
embedded in a porous manganese oxide
|
Using gold nanoparticles embedded in a porous manganese oxide as a room
temperature catalyst to breakdown volatile organic compounds in air.
|
|
A layer of closely spaced palladium
nanoparticles that detect
hydrogen. When hydrogen is absorbed the palladium nanoparticles swell, causing shorts between nanoparticles which lowers the resistance of
the palladium layer.
|
|
|
Quantum Dots (crystalline
nano particles)
|
Quantum Dots (crystalline
nanoparticles) that identify the location of cancer cells in the body.
|
|
Gold nano particles with organic molecules
|
Combining gold nanoparticles with organic
molecules to create a transistor known as a NOMFET (Nanoparticle Organic Memory Field-Effect Transistor).
|
|
Iron nano particles
|
Iron nanoparticles used to
clean up carbon tetrachloride pollution in ground water.
|
|
Silicon
nano particles coating
|
Silicon nanoparticles coating anodes of
lithium-ion batteries to increase battery power and reduce recharge time.
|
|
Gold nano particles
|
Gold nanoparticles that allow
heat from infrared lasers to be targeted on cancer tumours.
|
|
Silicate nano particles
|
Silicate nanoparticles
used to provide a barrier to gasses (for example oxygen), or moisture in a
plastic film used for packaging. This could reduce the possibly of food
spoiling or drying out.
|
|
Zinc oxide nano particles
|
Zinc oxide nano particles
dispersed in industrial coatings to protect wood, plastic and textiles from
exposure to UV rays.
|
|
Silicon dioxide
crystalline nano particles
|
Silicon dioxide crystalline nano particles
filling gaps between carbon fibres strengthen tennis racquets.
|
|
Silver nano particles
|
Silver nano particles
in fabric that kills bacteria making clothing odour-resistant.
|
|
Porous silica
Nano particles used to deliver chemotherapy
drugs to cancer cells.
|
Porous silica nano particles
used to deliver chemotherapy drugs to cancer cells.
|
|
Semiconductor
nano particles
|
Semiconductor nano particles applied in
a low temperature printing process that results in low cost solar cells.
|
|
Iron oxide nano particles
|
Iron oxide nano particles used to clean arsenic from water
wells.
|
|
|
Nano particles, when activated
by x-rays, that generate electrons that cause the
destruction of cancer cells to which they have attached themselves. This is
intended to be used in place radiation therapy with much less damage to
healthy tissue.
|
|
|
A nano particle cream that releases
nitric oxide gas to fight staph
infections.
|
|
Gold-palladium
|
That
can replace expensive and potentially toxic reagents that promote oxidation
of aromatic primary alcohols to aldehydes, which is one of the crucial
processes in the perfume production.
|
Showing posts with label crystal. Show all posts
Showing posts with label crystal. Show all posts
Tuesday, 24 February 2015
DIFFERENT NANO PARTICLES AND THEIR APPLICATIONS
Saturday, 20 September 2014
APPLICATIONS OF X-RAY DIFFRACTION
Because of the information it provides concerning the sub microscopic structure
of any kind of material from x-ray diffraction analysis, information usually
obtainable only by inference from other methods of examination, this method of
crystal analysis has become very important to modern industry. The large number
of industrial applications already made prove the value of these crystal structure
studies, and provide a firm foundation for this branch of technology. Hence the
chemist, the physicist, the metallurgist, and the engineer now have in x-ray
diffraction a powerful scientific instrument for use in the quest for better
methods and improved materials, and for the maintenance of required quality
throughout the manufacturing processes.
APPLICATIONS TO
METALLURGY AND METALLOGRAPHY
1. The
Composition and Structure of Alloys.
- Identification of alloy components and compounds.This is a special case of the general problem of chemical analysis by x-ray diffraction, and is used very frequently in many laboratories as a check on the results of other methods of examination.
- Differentiation between compound formation and solid solution.This is also a special case of chemical analysis in which a compound formed between two or more elements will give rise to a new x-ray pattern which is different from that of any of the constituents, while the solid solution will in general give the pattern of one of the elements, but with a shift in line positions which depends upon the relative amounts of the other elements present in the solution.
- Routine determination of percentage composition of solid solution phases, on the basis of measurement of line shift with varying amounts of solute present.
- Determination of the mechanism of alloy formation. This involves study of reflection and back reflection patterns of a series of alloys with various thermal treatments, and the correlation of the conclusions with chemical and microscopic data.
- Determination of miscibility limits and solid-phase boundaries of many-component alloy systems, by correlating lattice parameters with increasing percentage of alloying constituents.
- Working out and checking the details of the solidus phases of the equilibrium diagrams of binary and many-component alloy systems. X-ray diffraction analysis is the most convenient and dependable of the accepted modern methods for this purpose.
- The most rational classification of alloy types and systems has been made on the basis of x-ray crystal analysis.
- Study of the "order-disorder" phenomena in alloy systems.
2. The
Effects of Rolling and Working on Metals and Alloys.
- Determination of structural changes accompanying successive reductions of sheet and wire, as a comparison of methods of reduction .by different techniques.
- Study of the effect of initial grain size, carbon content, initial strip thickness, and of rolling variables on the final structure of rolled strip steel in determining the proper scientific methods of working and forming.
- Determination of the effects of twisting and bending strip and wire.
- Measurement of the extent of deformation and distortion by rolling, drawing, shaping, etc., as a routine check on the efficacy of the manufacturing process.
- Determination of slip planes, "fiber" structure, etc., of rolled sheet and drawn wires.
- Differentiation between surface and interior structures, or study of "zonal" structural characteristics.
- Determination of the most desirable structure of a sheet or wire to be subjected to a forming operation, and a rational method of classifying metals as to workability. This method is used in many laboratories to "grade" every production lot. In this way the sheet mill can guarantee delivery of metal best suited to the manufacturer's shaping processes,
- Furnishes an explanation of structural failures in spinning, cupping, and stamping operations. "Trouble shooting" in regard to these operations is one of the best paying uses of x-ray crystal analysis in the metallographic laboratory.
- Measurement of the depth of cold work caused by machining, drilling, punching, grinding, etc.
- Study of the mechanism of "fatigue" and other types-of metal failures, and in many cases a determination of the cause for premature or unexpected failures.
3. The
Effects of Annealing and Other Thermal Treatments on Metals.
- Establishment and routine maintenance of scientifically correct annealing techniques, and in many eases also for heat treating techniques.
- Study of recrystallization mechanism, and exact determination of recrystallization temperature.
- Study of precipitation and age hardening phenomena.
- Study of the relation of carbon content to annealing, and the relations between amount of reduction, time and temperature of anneal, and the final structure.
- Determination of quench and temper structures of spring steels, and a continuous check on hardening and tempering operations.
- Study of growth of texture in castings.
- Measurement of strain relief upon annealing.
- Determination of surface effects, such as decarburization, oxidation, excessive crystal growth, etc., as differentiated from interior structure.
4.
Miscellaneous Applications to Metals.
- Determination of true "crystal size" as distinguished from microscopic 'grain size". This is a common and much used procedure in many factories.
- Determination of the structure of welds and the presence of strain or distortion in the neighbourhood of the weld.
- Determination of the reason for and indication of the cure for "embrittlement" of malleable iron.
- Measurement of crystal size, crystal orientation, and absence of distortion (or degree of crystal perfection) in relation to electrical and magnetic properties of transformer steels.
- Determination of the effects of thermal treatments on the "spoilage" and recovery of permanent magnet alloys.
- Determination of uniformity, depth, and mechanism of surface hardening.
- Measurement of crystal size, preferred orientation, and thickness of electrodeposited films, a routine check on the plating process.
- Determination of the chemical composition of protective films, and study of mirrors and sputtered films.
- Study of the effects of included and absorbed or adsorbed gases on the structure of metals.
- Determination of optimum crystal size and best structure for electrical contact points, and a continuous check on these during manufacture.
- Study of the effects of crystal size and crystal orientation on electrical properties.
- Aid in the study of corrosion and corrosion or thermal "fatigue" and chemical embrittlement, and determination of the chemical composition of boiler scales.
- Furnishes a scientific approach to the preparation of new alloys, and a prediction of the properties of new or untried alloys.
- Study of the transition zone between base and covering of plated or enameled metals.
- Rational determination of the effects of minute impurities upon the structure of metals.
- Identification of inclusions in metals. This is a special case of chemical analysis by x-ray diffraction.
- An absolute and non-destructive measure of residual elastic surface stresses in metals. This is used quite extensively in several countries in the study of steel structures such as bridges and building frameworks.
- Determination of particle size in the colloidal region.
APPLICATIONS IN
CHEMISTRY
1. General
and Physical Chemistry
- Determination of ultimate crystal structure, including lattice types, unit cell dimensions, atomic positions, ionic groupings, and crystallographic systems of substances.
- Furnishes a unique and unquestionable characterization of individual chemical compounds. This is the basis of the wide-spread use of x-ray diffraction for chemical analysis. The analysis is, of course, made in terms of chemical compounds rather than in terms of elements and ionic groupings.
- Differentiation between a mixture, solid solution or complex compound formation.
- Supplies a quantitative estimate of the relative amounts of the various compounds in a mixture. The estimate can be refined by the proper use of a recording microphotometer.
- Furnishes a certain test for the crystallinity or non-crystallinity of a material, either in the solid state or in solution.
- Determination of crystal sizes in the microscopic and sub-microscopic (colloidal) ranges.
- Study of allotropic modifications and transitions of an element or compound, and the effects of impurities on these.
- Determination of the ideal or theoretical density of a substance, giving a basis for the estimation of porosity.
- Differentiation between true and false hydrates.(Chemical analysis.)
- Discovery of unsuspected chemical reactions.
- Recognition of colloidally dispersed phases, and differentiation between true solutions and suspensions.
- Determination of crystal size and structure of colloidal so is and gels.
- Identification of adsorbed films and chemical changes involved in adsorption.
- Determination of optimum crystal sizes and orientations for maximum catalytic activity, and study of the mechanism of catalysis and "poisoning" of catalysts. This is used not only to find the best processes for preparing a catalyst but also as a routine test of production.
- Determination of molecular sizes in liquid solutions, and molecular weights of liquids.
- Determination of the mechanism and course of dry reactions and allotropic transformations in the solid state, even at extremely high or extremely low temperatures.
2. Organic
Chemistry
The list given above for General and Physical Chemistry, and in addition
furnishes:
- A sure test for the identity or non-identity of synthetic and naturally occurring materials.
- Estimation of molecular weights of hydrocarbons, etc.
- Measurement of atomic sizes, interatomic distance and diameters of molecules.
- A method of following chemical reactions, as for example addition to or oxidation of a multiple bond.
- Estimation of the purity of soaps, acids, etc.
- Estimation of the positions of side chains and functional groups.
- Measurement of the thickness of oriented films.
- Determination of molecular orientation in fibers, and molecular structure of naturally occurring fibers and membranes.
- A method of following polymerization and condensation reactions, and decomposition in breaking up long chain compounds.
- Study of lubrication and lubricants, including a routine method of quantitatively comparing efficiency of lubricants.
- Study of changes taking place in the ripening of cheese, and during other processing of dairy products.
- A rational classification of synthetic and natural plastics, and a qualitative scheme for identification of these.
3.
Analytical Chemistry.
In addition to the applications listed above, x-ray diffraction provides
for:
- Identification of the chemical composition of precipitates.
- Tests for purity and identification of impurities in precipitates.
- Measurement of particle (crystal) sizes of precipitates in relation to treatment and reagent concentrations.
- Determination of the state of perfection of the crystal lattice in precipitates, particularly in regard to aging effects, etc.
APPLICATIONS IN
THE PROCESS INDUSTRIES
Since the process industries are engaged in chemical manufacture, the general
applications listed under "chemistry" could be repeated here. To avoid
duplication, however, only those applications of x-ray crystal analysis to some
particular problems will be given.
1. Paints
and Pigments.
- Structure and crystal sizes as functions of color, spreading, wetting and obscuring power, stability, gloss, and method of preparation.
- Study of the drying and setting of oils, the mechanisms of the reactions involved, etc., and their relationships to the structure and composition of pigments.
- Tests for solution of driers, and study of the mechanisms of their action.
- Routine analysis for purity of pigments. This is an important production test, particularly for those pigments which can exist in more than one crystal form, as for example titanium dioxide.
2. Ceramics
and Glass.
- Routine qualitative and quantitative analysis of materials and clay mixtures, in terms of compounds present.
- Determination of the structural and chemical changes occurring during sintering, fusing, and other thermal treatments and the mechanisms of these reactions.
- Furnishes the best and fastest method for determining and checking the solidus phases of many component systems, and for determining miscibility limits.
- Gives a definite test for incipient devitrification of glass.
- Identification of substances imparting color or opacity to glasses or enamels.
- Determination of crystal size with relation to color of pigment.
- Study of transition zones between base metal and vitreous enamel.
- Measurement of chemical reaction rates in melt or during sintering.
3. Cement
and Plaster.
- Study of reaction rates and mechanisms taking place during manufacture and use of cement.
- Routine chemical analysis of raw materials and clinker.
- Differentiation between particle size of aggregates and true crystal size.
- Method of determining and checking complex phase diagrams with certainty.
- Investigation of setting accelerators and their effects on the final structure of concrete.
- Control analysis of lime for crystal size, etc., to ensure proper plastic properties of plaster.
- Study of structure of limestone and its kiln behaviour in relation to the properties of the final product.
- Study of the dehydration of gypsum and the structural changes involved in the use and reuse of plaster of Paris molds.
4. Storage
Batteries.
- Study of physical and chemical structure of plates as related to performance.
- Study of chemical reactions occurring during charge and discharge.
- Study of the influence of the structure of grid and composition and aging of the paste upon the physical properties of the plates, and control analysis for the manufacturing process.
- Identification of deposits and sediments on plates, separators, and in cell.
5. Rubber
and Allied Products.
- Study of chemical reactions taking place during vulcanization and other processing.
- Determination of crystallinity, state of dispersions, crystal sizes of fillers, etc., and their relation to the physical characteristics of the finished products.
- Study of the basic structure of rubber and rubber-like materials. X-ray diffraction furnishes the only sure test of the fundamental relationships between natural and synthetic rubber.
- Study of fabrics and other binding materials used in the manufacture of rubber products, and routine grading of fibers as explained below.
6. Textiles
and Fibers.
- Determination of the degree'of fiberingn . A quantitative relationship between the degree of fibering and tensile strength of cotton fibers has been developed and is being used as a routine method of grading cotton.
- Furnishes a scientific method of classifying cotton, silk, wool, and other natural and synthetic fibers.
- Determination of the rate, mechanism, and completeness of mercerization, nitration, and other chemical reactions, and use in control analysis.
- Determination of the mechanism of fire-proofing fibers, and of exact amount of reagent required.
- Identification of adsorbed films and the chemical changes involved in adsorption, particularly as applied to dyeing of fibers.
- Great improvements in quality, tensile strength, and non-wrinkling properties of rayon and other synthetic fibers has been made through x-ray studies. The development of artificial wool from skim milk, peanuts, beans, etc., can be traced directly to x-ray diffraction studies of the structures of the various proteins. The development of "nylon", the new synthetic silk, has depended to a great degree on x-ray studies of its fiber characteristics by x-ray diffraction.
- X-ray diffraction studies on collagen fibers (side walls of animal intestines, tendons, etc.) have resulted in enormous improvement in the quality and wearing properties of tennis racket strings, and in the strength and controlled digestibility of surgical ligatures and sutures.
APPLICATIONS IN MINERALOGY
1. General
Mineralogy.
- Complete and unambiguous mineralogical analysis of ores, clays, and other mineral mixtures.
- Analysis of industrial dusts, and correlation with the occurrence of industrial diseases.
- Classification and evaluation of certain commercial ores.
- Identification and classification of the clay minerals and complexes making up the so-called soil-colloid.
- A scientific method of studying the changes produced in natural minerals by weathering, accelerated weathering tests, and other chemical and physical degradations.
- Specifications for asbestos, mica, and other natural insulating materials for special purposes.
- Classification of coal, charcoal, etc.
2. Precious
Stones and Gems.
- Identification, classification, and differentiation of genuine, both natural and synthetic, and imitation gems by a non-destructive test.
- Differentiation between natural and synthetic gems, nondestructively.
- Differentiation between natural and cultured pearls, non-destructively. This is a routine procedure with some of the leading jewelry manufacturers throughout the world.
- Determination of the proper orientation for a "jeweled" bearing (in watches, electric meters, etc.) to give maximum service and wearing qualities.
- Selection and classification of "black" diamonds for drills and dies, determination of causes for undue wear, and proper crystallographic orientations for optimum service.
- Determination of the proper direction of cutting quartz crystals for crystal oscillators in radio broadcasting and telephone equipment.
APPLICATIONS IN
PHYSIOLOGY, PATHOLOGY, AND BIOLOGY
1. The
applications under this heading are quite recent developments and are not yet
generally used. Listing of some, however, will serve to show the general trend
and possibilities of x-ray diffraction research in these complex and difficult,
but extremely important fields,
- Differentiation between some normal and pathological tissues.
- Study of the effects of diseases on the structures of tissues, as on bone structure changes in rickets, cancer of the bone, and other bone diseases.
- Study of structure of living tissue, as nerve and muscle, in relation to body functions.
- Identification and classification of mineral deposits in organs, such as calcifications, gall stones, siliceous deposits, etc.Much interest is evident at present in the study of the action of free quartz on lung tissue in silicosis, and of other industrial diseases and their occurrence, and many papers have been published in medicinal journals on x-ray diffractipn studies of silicotic lung tissue.
- Structure and classification of tooth enamel, dentyne, etc., and structures of the teeth in relation to diet.
2. Papers of
interest to pharmacists have appeared recently on the following subjects:
- Identification of minerals in rhubarb.
- Differentiation between natural and synthetic camphor.
- Study of the reactions between menthol and the mercuric oxides.
Wednesday, 4 December 2013
WAVELENGTH DISPERSIVE X-RAY FLUORESCENCE (WD-XRF)
INTRODUCTION
Wavelength Dispersive X-Ray Fluorescence Spectrometry (WD-XRF) is the oldest method of measurement of X-rays, introduced commercially in the 1950’s. This name is descriptive in that the radiation emitted from the sample is collimated with a Soller collimator, and then impinges upon an analyzing crystal. The crystal diffracts the radiation to different extents, according to Bragg’s law, depending upon the wavelength or energy of the X radiation. This angular dispersion of the radiation permits the sequential or simultaneous detection of X-rays emitted by elements in the sample. Simultaneous instruments normally contain several sets of analyzing crystals and detectors; one is adjusted for each desired analyte in the sample. These instruments tend to be very expensive, but efficient for the routine determination or preselected elements.
WD-XRF spectrometers are usually larger and more expensive than other spectrometers. Because the analyzing crystal d-spacing determines wavelength sensitivity, they are usually more sensitive than other spectrometers. To overcome losses in X-ray optics of the WD-XRF spectrometers and to maximize primary radiation intensity, X-ray tubes are usually employed. The sample is usually held under vacuum to reduce contamination and avoid absorption of light element characteristic radiation in air.
Typical uses of WD-XRF include the analysis of oils and fuel, plastics, rubber and textiles, pharmaceutical products, foodstuffs, cosmetics and body care products, fertilizers, minerals, ores, rocks, sands, slags, cements, heat-resistant materials glass, ceramics, semiconductor wafers; the determination of coatings on paper, film, polyester and metals; the sorting or compositional analysis of metal alloys, glass and polymeric materials; and the monitoring of soil contamination, solid waste, effluent, cleaning fluids, sediments and air filters.
PRINCIPLE OF WD-XRF
WD-XRF spectrometers measure X-ray intensity as a function of wavelength. This is done by passing radiation emanating from the specimen through an analyzing diffraction crystal mounted on a 2θ goniometer. By Bragg’s Law, the angle between the sample and detector yields the wavelength of the radiation:
2d sin θ = nλ ;
where:
d is the d-spacing of the analyzing crystal,
θ is half the angle between the detector and the sample,
n is the order of diffraction.
The analyzing crystal must be oriented so that the crystal diffraction plane is directed in the appropriate direction. Figure shows a simplified schematic of the WD-XRF spectrometer. A scintillation or flow-proportional detector usually measures the fluoresced radiation. The heights of the resulting pulses are proportional to energy so using a pulse
1. COLLIMATOR MASKS
The collimator masks are situated between the sample and collimator and serve the
purpose of cutting out the radiation coming from the edge of the cup aperture (Figure 2).
The size of the mask is generally adapted to suit of the cup aperture being used.
The masks perform one of the two functions: background reduction and improved
fluorescence (Figure 3).
 |
2.Use of Cu 200 μm filter for cutting off the radiation coming from Rh X-ray tube.
|
 |
3. Use of Al 100 μm filter for improvement of the ratio peak/background.
|
2. COLLIMATOR
 |
4. Collimators with different angles of resolution.
|
Collimators consist of a row of parallel slats (Figure 4) and select a parallel beam of X-rays coming from the sample and striking the crystal. The spaces between the slats determine the degree of parallelism and thus the angle resolution of the collimator.
A 0.077° collimator is adequate for high resolution measurement parameters. Collimators with low resolution (e.g. 1.5 -2.0°) are advantageous for light elements such as Be, B and C (Figure 5). Using a collimator with a low resolution increases then intensity significantly. This enables intensity to be increased without a loss in angle resolution when analyzing light elements.
 |
5. Example of the influence of collimator resolution on the intensity of a light
element.
|
3. THE ANALYZING CRYSTALS
a. Bragg’s Law
Crystals consist of a periodic arrangement of atoms (molecules) that form the crystal lattice. In such an arrangement of particles you generally find numerous planes running in different directions through the lattice points (= atoms, molecules), and not only horizontally and vertically but also diagonally. These are called lattice planes. All of the planes parallel to a lattice plane are also lattice planes and are at a defined distance from each other. This distance is called the lattice plane distance “d”.
 |
| 6. Bragg’s Law. |
When parallel X-ray light strikes a lattice plane, every particle within it acts as a scattering center and emits a secondary wave. All of the secondary waves combine to form a reflected wave. The same occurs on the parallel lattice planes for only very little of the X-ray wave is absorbed within the lattice plane distance “d”. All these reflected waves interfere with each other. If the amplification condition “phase difference = a whole multiple of the wavelength” (Δλ = nλ) is not precisely met, the reflected wave will interfere such that cancellation occurs. All that remains is the wavelength for which the amplification condition is met precisely. For a defined wavelength and a defined lattice plane distance, this is only given with a specific angle, the Bragg angle (Figure 6).
Under amplification conditions, parallel, coherent X-ray light (1,2) falls on a crystal
with a lattice plane distanced ‘d’ and is scattered below the angle θ (1′,2′). The proportion of the beam that is scattered on the second plane has a difference of ‘ACB’ to the proportion of the beam that was scattered at the first plane. The amplification condition is fulfilled when the phase difference is a whole multiple of the wavelength λ. This results in Bragg’s Law:
2d sin θ = nλ ;
n = 1, 2, 3… Reflection order.
On the basis of Bragg’s Law, by measuring the angle θ, we can determine either the
wavelength λ, and thus chemical elements, if the lattice plane distance ‘d’ is known or, if the wavelength λ is known, the lattice plane –value distance ‘d’ and thus the crystalline structure.
This provides the basis for two measuring techniques for the quantitative and qualitative determination of chemical elements (XRF) and crystalline structures (molecules, XRD), depending on whether the wavelength λ or the 2d-value is identified by measuring the angle θ.
- In X-ray diffraction (XRD) the sample is excited with monochromatic radiation of a known wavelength (λ) in order to evaluate the lattice plane distance as per Bragg’s equation.
- In XRF, the ‘d’-value of the analyzer crystal is known and we can solve Bragg’s equation for the element characteristic wavelength (λ).
b. Reflections of Higher Orders
Figures 7a and 7b illustrate the reflections of the first and second order of one wavelength below the different angles θ1 and θ2. Here, the total reflection is made up of the various reflection orders (1, 2 …, n). The higher the reflection order, the lower the intensity of the reflected proportion of radiation generally is. How great the maximum detectable order is depends on the wavelength, the type of crystal used and the angular range of the spectrometer.
 |
| 7 a. First order reflection: λ = 2 d sin θ1. |
 |
| 7 b. Second order reflection: 2λ = 2 d sin θ2. |
It can be seen from Bragg’s equation that the product of reflection orders ‘n = 1; 2; ..’
and wavelength ‘λ’ for greater orders, and shorter wavelengths ‘λ* < λ’ that satisfy the
condition ‘λ* = λ/n’, give the same result.
Accordingly, radiation with one half, one third, one quarter etc. of the appropriate
wavelength (using the same type of the crystal) is reflected below the identical angle θ:
1λ = 2(λ / 2) = 3(λ / 3) = 4(λ / 4) = ……......
As the radiation with one half of the wavelength has twice the energy, the radiation with one third of the wavelength three times the energy etc., peaks of twice, three times the energy etc. can occur in the pulse height spectrum (= energy spectrum) as long as appropriate radiation sources.
c. Crystal Types
The wavelength dispersive X-ray fluorescence technique can detect every element
above the atomic number 4 (Be). The wavelengths cover the range of values of four
magnitudes: 0.01 – 11.3 nm. As the angle θ can theoretically only be between 0° and 90° (in practice 2° to 75°), sinθ an only accept values between 0 and +1. When Bragg’s equation is applied:
0 < nλ/2d = Sinθ < 1
This means that the detectable element range is limited for a crystal with a lattice plane difference ‘d’. Therefore a variety of crystal type with different ‘2d’ values is necessary to detect the whole element range (from atomic number 4).
Besides the ‘2d’ values, the following selection criteria must be considered when a
particular type of crystal is to be used for a specific application:
- Resolution
- Reflectivity (→ intensity)
Further criteria can be:
- Temperature stability
- Suppression of higher orders
- Crystal fluorescence.
d. Dispersion, Line Separation
The extent of the change in angle Δθ upon changing the wavelength by the amount Δλ (thus: Δθ/Δλ) is called “dispersion”. The greater the dispersion, the better is the separation of two adjacent or overlapping peaks. Resolution is determined by the dispersion as well as by surface quality and the purity of the crystal.
Mathematically, the dispersion can be obtained from the differentiation of the Bragg
equation.
Δθ /Δλ = n/2dsinθ
It can be seen from this equation that the dispersion (or peak separation) increases as the lattice plane distance ‘d’ declines.
e. Synthetic Multilayers
 |
8. Diffraction in the layers (here: Si/W) of a multilayer.
|
Multilayers are not natural crystals but artificially produced ‘layer analyzers’. The lattice plane distances ‘d’ are produced by applying thin layers of two materials in alternation on to a substrate (Figure 8). Multilayers are characterized by high reflectivity and a somewhat reduced resolution. For the analysis of light elements the multilayer technique presents an almost revolutionary improvement for numerous applications in comparison to natural crystals with large lattice plane distances.
4. DETECTORS
When measuring X-ray, use is made of their ability to ionize atoms and molecules, i.e. to displace electrons from their bonds by energy transference. In suitable detector materials, pulses whose strengths are proportional to the energy of the respective X-ray quants are produced by the effect of X-ray. The information about the X-ray quarts energy is contained in the registration of the pulse height. The number of X-ray quants per unit of time, e.g. pulses per second (cps = counts per second, KCps = kilocounts per second), is called their intensity and contains in a first approximation the information about the concentration of the emitting in the sample. Two main types of detectors are used in wavelength dispersive X-ray fluorescence spectrometers: the gas proportional counter and the scintillation counter.
a. Gas Proportional Counter
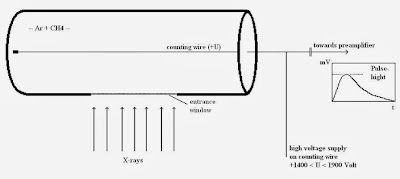 |
| 9: A gas proportional counter. |
The gas proportional counter comprises a cylindrical metallic tube in the middle of which a thin wire (counting wire) is mounted. This tube is filled with a suitable gas (e.g. Ar+ 10% CH4). A positive high voltage (+U) is applied the wire. The tube has a lateral aperture or window that is sealed with a material permeable to X-ray quants (Figure 9).
An X-ray quant penetrates the window into the counter’s gas chamber where it is absorbed by ionizing the gas atoms and molecules. The resultant positive ions move to the cathode (tube), the free electrons to the anode, the wire. The number of electron-ion pairs created is proportional to the energy of the X-ray quant. To produce an electron-ion pair,approx. 0.03 keV are necessary, i.e. the radiation of the element boron (0.185 keV) produces approx. 6 pairs and the K-alpha radiation of molybdenum (17.5 keV) produces approx. 583 pairs. Due to the cylinder geometric arrangement, the primary electrons created in this way see an increasing electrical field on route to the wire.
The high voltage in the counting tube is now set so high that the electrons can obtain enough energy from the electrical field in the vicinity of the wire to ionize additional gas particles. An individual electron can thus create up to 10.000 secondary electron-ion pairs. The secondary ions moving towards the cathode produce measurable signal. Without this process of gas amplification, signals from boron, for example, with 6 or molybdenum with 583 pairs of charges would not be able to be measured as they would not be sufficiently discernible from the electronic noise. As amplification is adjustable via high voltage in the counting tube and is set higher for measuring boron than for measuring molybdenum. The subsequent pulse electronics supply pulses of voltage whose height depends, amongst other factors, on the energy of the X-ray quants.
b. Scintillation Counters
 |
10. Scintillation counter including photomultiplier.
|
The scintillation counter, “SC”, used in XRF comprises a sodium iodide crystal in which thallium atoms are homogeneously distributed ‘NaI(Tl)’. The density of the crystal is sufficiently high to absorb all the XRF high energy quants. The energy of the pervading X-ray quants is transferred step by step to the crystal atoms that then radiate light and cumulatively produce a flash. The amount of light in this cintillation flash is proportional to the energy that the X-ray quant has passed to the crystal. The resulting light strikes a photocathode from which electrons can be detached very easily. These electrons are accelerated in a photomultiplier and, within an arrangement of dynodes, produce so-called secondary electrons giving a measurable signal once they have become a veritable avalanche (Figure 10). The height of the pulse of voltage produced is, as in the case of the gas proportional counter, proportional to the energy of the detected X-ray quant.
c. Pulse Height Analysis (PHA), Pulse Height Distribution
If the number of the measured pulses (intensity) dependent on the pulse height is displayed in a graph, we have the ‘pulse height spectrum’. Synonymous terms are: ‘pulse height analysis’ or ‘pulse height distribution’. As the height of the pulses of voltage is proportional to the X-ray quants energy, it is also referred to as the energy spectrum of the counter (Figure 11a and 11b). The pulse height is given in volts, scale divisions or in ‘%’ (and could be started in keV after appropriate calibration). The “%”-scale is defined in such a way that the peak to be to be analyzed appears at 100 %.
 |
11.a. Pulse height distribution
|
 |
11.b . Pulse height distribution (Fe) Scintillation counter.
|
 |
12. Pulse height distribution (Fe) with escape peak.
|
If argon is used as the counting gas component in gas proportional counters, an
additional peak, the escape peak (Figure 12), appears when X-ray energies are irradiated that are higher than the absorption edge of argon.
The escape peak arises as follows:
The incident X-ray quant passes its energy to the counting gas thereby displaying a K electron from an argon atom. The Ar atom can now emit an Ar Kα1,2 X-ray quant with an energy of 3 keV. If this Ar-fluorescence escapes from the counter then only the incident energy minus 3 keV remains for the measured signal. A second peak, the escape peak that is always 3 keV below the incident energy, appears in the pulse height distribution.
When using other counting gases (Ne, Kr, Xe) instead of argon, the escape peaks appear with an energy difference below the incident energy that is equivalent to the appropriate emitted fluorescence radiation (Kr, Xe). Using neon as the counting gas component produces no recognizable escape peak as the Ne K-radiation, with energy of 0.85 keV, is almost completely absorbed in the counter. Also, the energy difference to the incident of 0.85 keV and the fluorescence yield are very small.
Subscribe to:
Posts (Atom)
